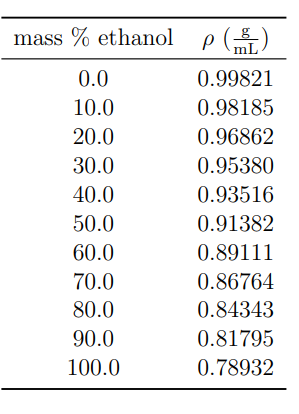
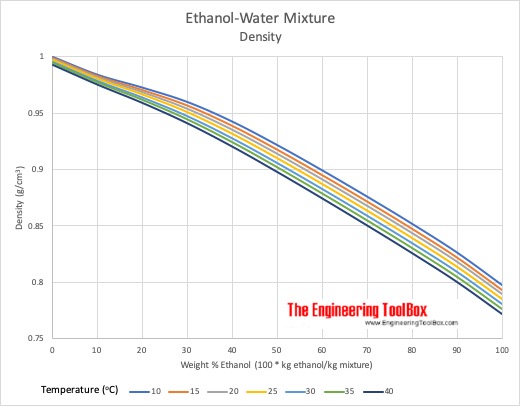
Computing the Composition of Ethanol-Water Mixtures Based on Experimental Density and Temperature Measurements
Density and Thermal Expansion of Ethyl Alcohol and of its Mixtures with Water - Page 424 - UNT Digital Library
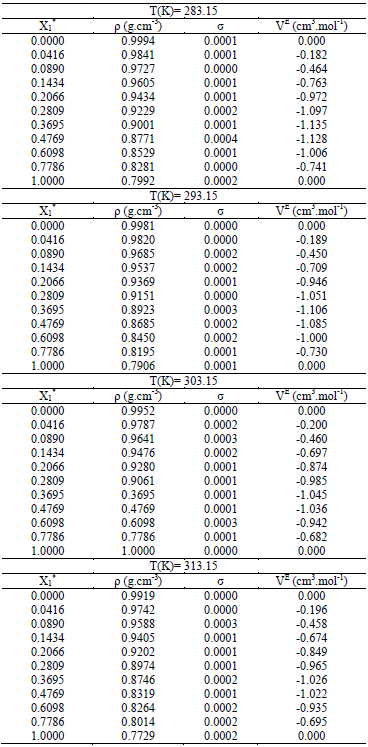
Densities and excess molar volumes for binary solution of water + ethanol, + methanol and + propanol from (283.15 TO 313.15) K

The density of water and ethanol at room temperature is 1.0 g / mL and 0.789 g / mL respectively. What volume of ethanol contains the same number of molecules as are

Calculate the molarity of a solution of ethanol in water in which the mole fraction of ethanol is 0.040 (assume the density of water to be one). Use the data given in

SOLVED: 6) Calculate the average density of the unknown liquid: average density of unknown liquid artrag %f uuknown liqui rdlume of unknoun liqund (mL) c) Use the known densities of ethanol and
Computing the Composition of Ethanol-Water Mixtures Based on Experimental Density and Temperature Measurements
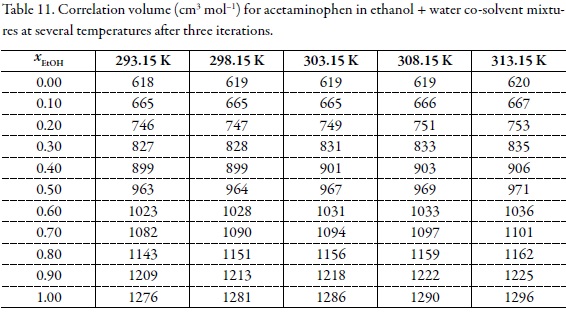
Preferential solvation of acetaminophen in ethanol + water solvent mixtures according to the inverse Kirkwood-Buff integrals method
SOLVED: (Apts) Conclusions Use the known densities of ethanol and 20% NaCl solution given in the table below to identify your unknown Iiquic from Part !. Solution Density (g/mL) ethanol 0.789 20%