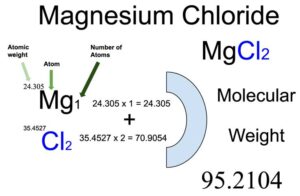
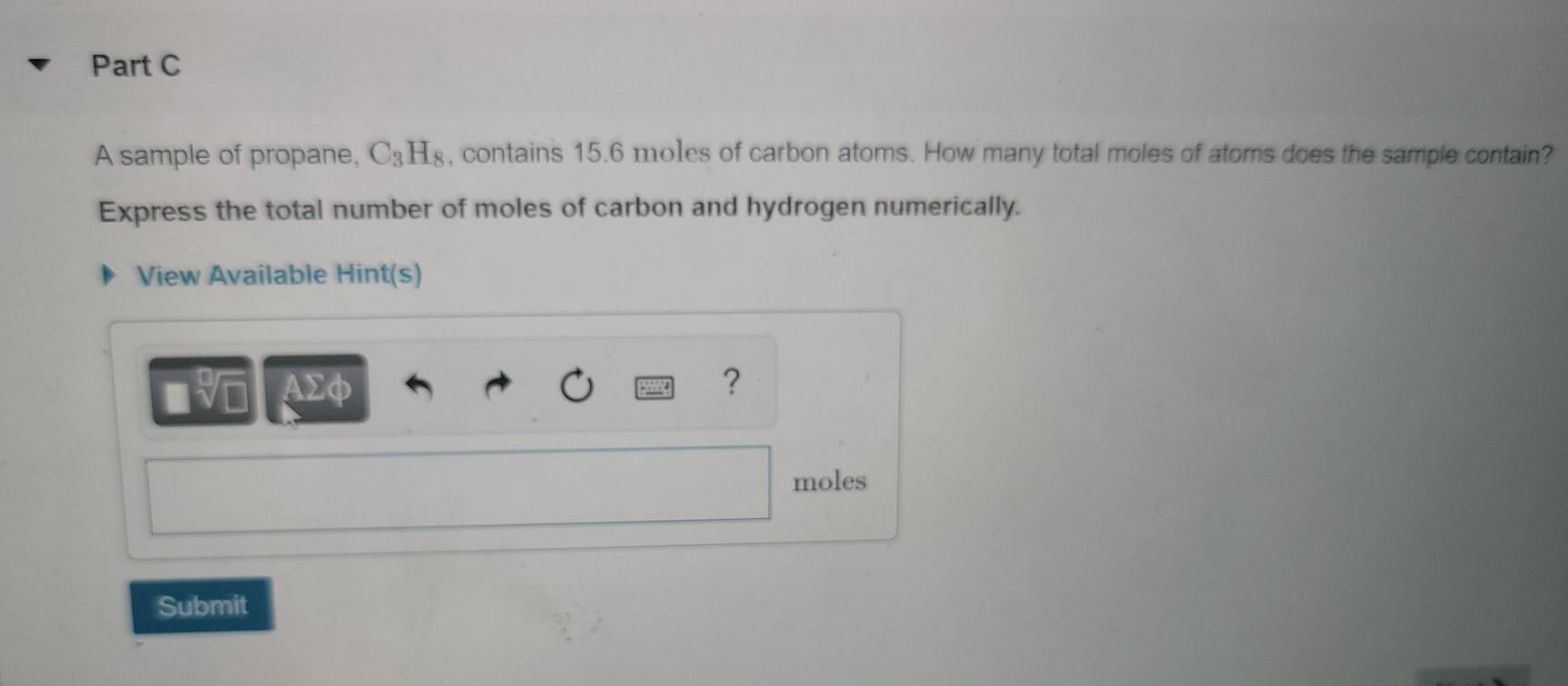
13.48 Calculate the number of moles of solute present in each of the following aqueous solutions: 185 mL of 1.50 M HNO mg that is 1.25 m NaCl. - ppt video online download
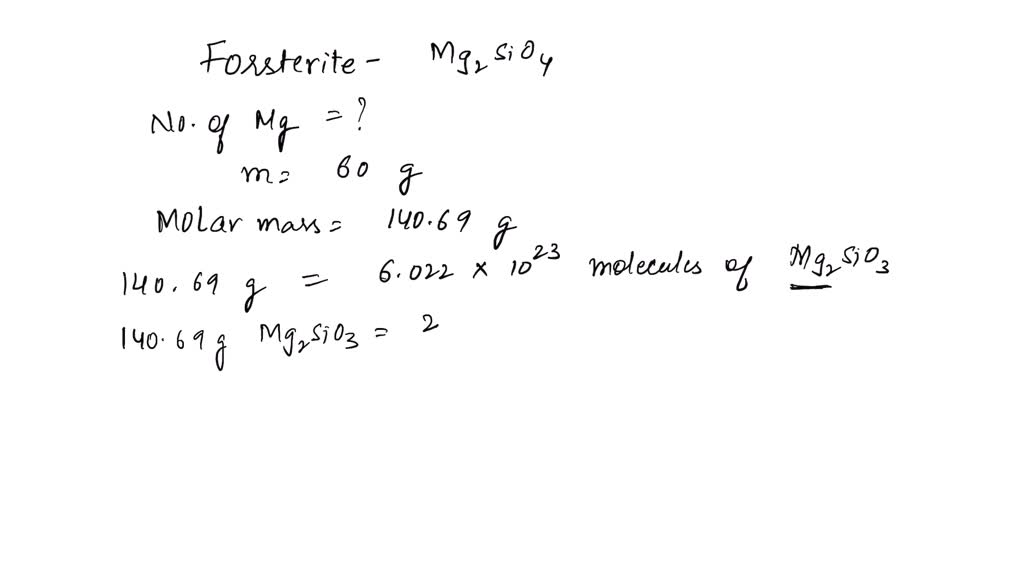
SOLVED: Calculate the number of magnesium atoms in a 60.0g sample of forsterite . Be sure your answer has a unit symbol if necessary, and round it to significant digits.
![Whole-rock mg-number [(Mg/(Mg + Fe 2+ ) × 100] vs olivine composition... | Download Scientific Diagram Whole-rock mg-number [(Mg/(Mg + Fe 2+ ) × 100] vs olivine composition... | Download Scientific Diagram](https://www.researchgate.net/profile/Michael-Garcia-15/publication/237505959/figure/tbl3/AS:393280111038464@1470776693021/Microprobe-analyses-of-plagioclase-in-two-samples-from-vent-D-episode-54_Q320.jpg)
Whole-rock mg-number [(Mg/(Mg + Fe 2+ ) × 100] vs olivine composition... | Download Scientific Diagram
![Whole-rock mg-number [(Mg/(Mg + Fe 2+ ) × 100] vs olivine composition... | Download Scientific Diagram Whole-rock mg-number [(Mg/(Mg + Fe 2+ ) × 100] vs olivine composition... | Download Scientific Diagram](https://www.researchgate.net/profile/Michael-Garcia-15/publication/237505959/figure/fig3/AS:668288493301765@1536343801912/Whole-rock-mg-number-Mg-Mg-Fe-2-100-vs-olivine-composition-in-forsterite_Q640.jpg)
Whole-rock mg-number [(Mg/(Mg + Fe 2+ ) × 100] vs olivine composition... | Download Scientific Diagram
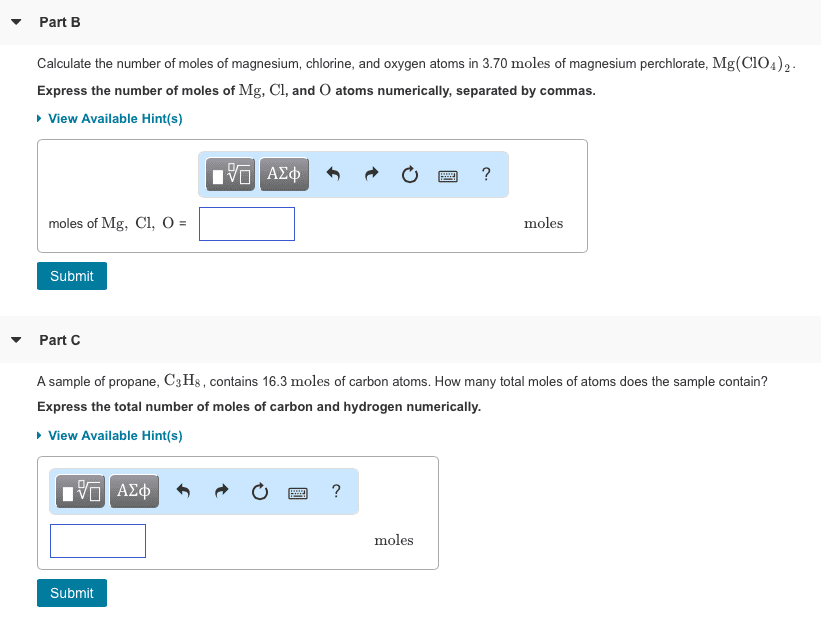
OneClass: Part B Calculate the number of moles of magnesium, chlorine, and oxygen atoms in 3.70 moles...
![Whole-rock mg-number [(Mg/(Mg + Fe 2+ ) × 100] vs olivine composition... | Download Scientific Diagram Whole-rock mg-number [(Mg/(Mg + Fe 2+ ) × 100] vs olivine composition... | Download Scientific Diagram](https://www.researchgate.net/publication/237505959/figure/fig3/AS:668288493301765@1536343801912/Whole-rock-mg-number-Mg-Mg-Fe-2-100-vs-olivine-composition-in-forsterite.png)
Whole-rock mg-number [(Mg/(Mg + Fe 2+ ) × 100] vs olivine composition... | Download Scientific Diagram
![Calculate the number of molecules present in 0.5 moles of magnesium oxide (MgO) . [Atomic weights : Mg = 24, O = 16 ] Calculate the number of molecules present in 0.5 moles of magnesium oxide (MgO) . [Atomic weights : Mg = 24, O = 16 ]](https://dwes9vv9u0550.cloudfront.net/images/2527137/b4ebf9c0-6b64-43b4-94e9-555f3180cc58.jpg)
Calculate the number of molecules present in 0.5 moles of magnesium oxide (MgO) . [Atomic weights : Mg = 24, O = 16 ]

calculate the number of atoms in 48g of magnesium - Science - Atoms and Molecules - 13145601 | Meritnation.com

Calculate the number of moles of magnesium oxide, MgO in i. 80 g and ii. 10 g of the compound. (Average atomic masses of Mg = 24 and O = 16 )
Calculate the number of particles in each of the following: (a) 48 g of Mg (b) 8 g of O2 (c) 0.1 mole of carbon (Atomic mass Mg = 24 u, O =
The chlorophyll responsible for photosynthesis contains 2.68% Mg by mass. What is the total number of magnesium atoms present in 2 g of it? - Quora