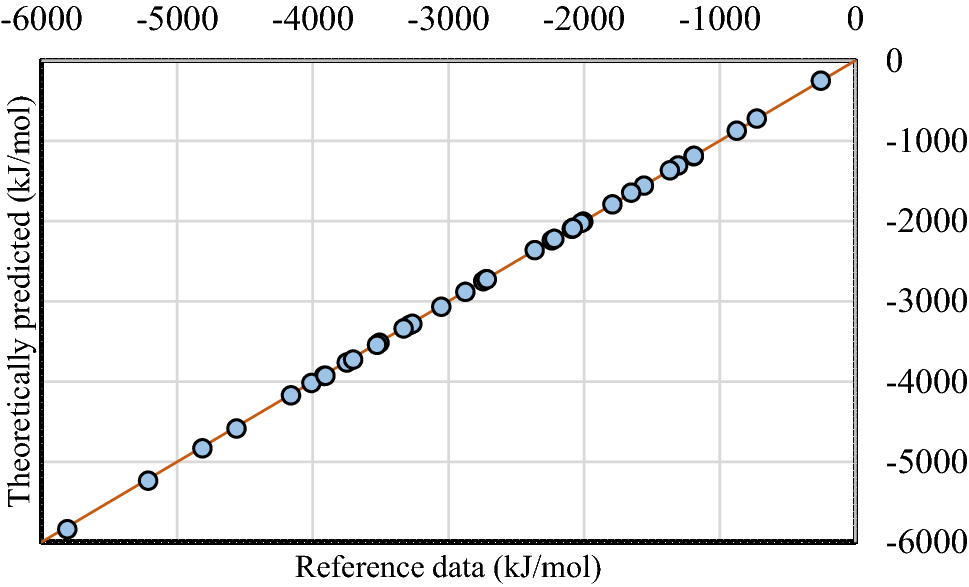
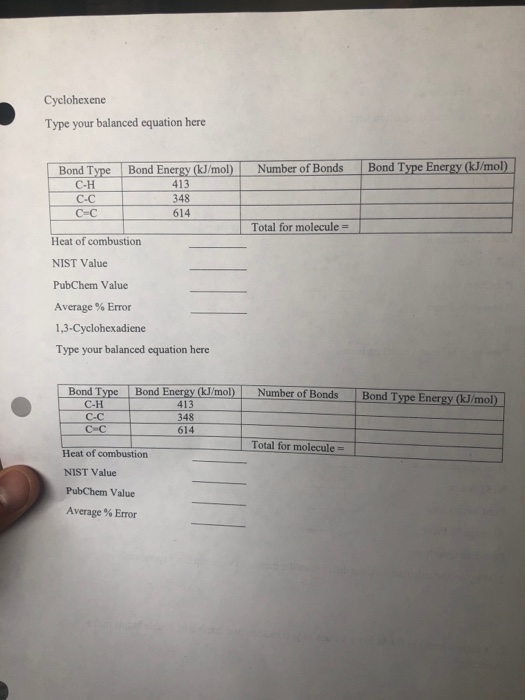
High-Level Quantum Chemistry Reference Heats of Formation for a Large Set of C, H, N, and O Species in the NIST Chemistry Webbook and the Identification and Validation of Reliable Protocols for

Gibbs free energy of formation for MTSg in the JANAF Thermochemical... | Download Scientific Diagram
The IUPAC-NIST Solubility Data Series: A Guide to Preparation and Use of Compilations and Evaluations: Journal of Physical and Chemical Reference Data: Vol 39, No 2
![PDF] Selected Values of Chemical Thermodynamic Properties. Tables for the First Thirty-Four Elements in the Standard Order of Arrangement. | Semantic Scholar PDF] Selected Values of Chemical Thermodynamic Properties. Tables for the First Thirty-Four Elements in the Standard Order of Arrangement. | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/fdbfea91525e122bc3146e0103077d28a16c564a/6-Table1-1.png)
PDF] Selected Values of Chemical Thermodynamic Properties. Tables for the First Thirty-Four Elements in the Standard Order of Arrangement. | Semantic Scholar

High-Level Quantum Chemistry Reference Heats of Formation for a Large Set of C, H, N, and O Species in the NIST Chemistry Webbook and the Identification and Validation of Reliable Protocols for

First law and Second law of thermodynamics applied to combustion - Part 2 - Enthalpy Calculation - YouTube

Heat of formation of aluminum fluoride by direct combination of the elements - Journal of Research of NIST and Predecessor Publications - NIST Digital Archives
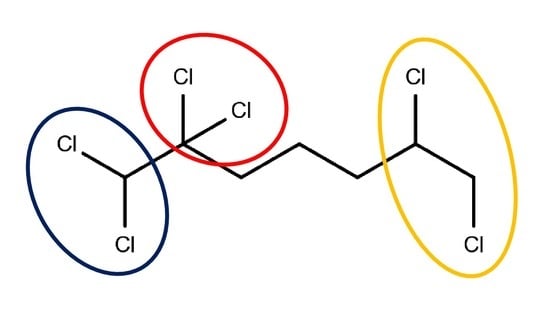
AppliedChem | Free Full-Text | Group Contribution Revisited: The Enthalpy of Formation of Organic Compounds with “Chemical Accuracy” Part III
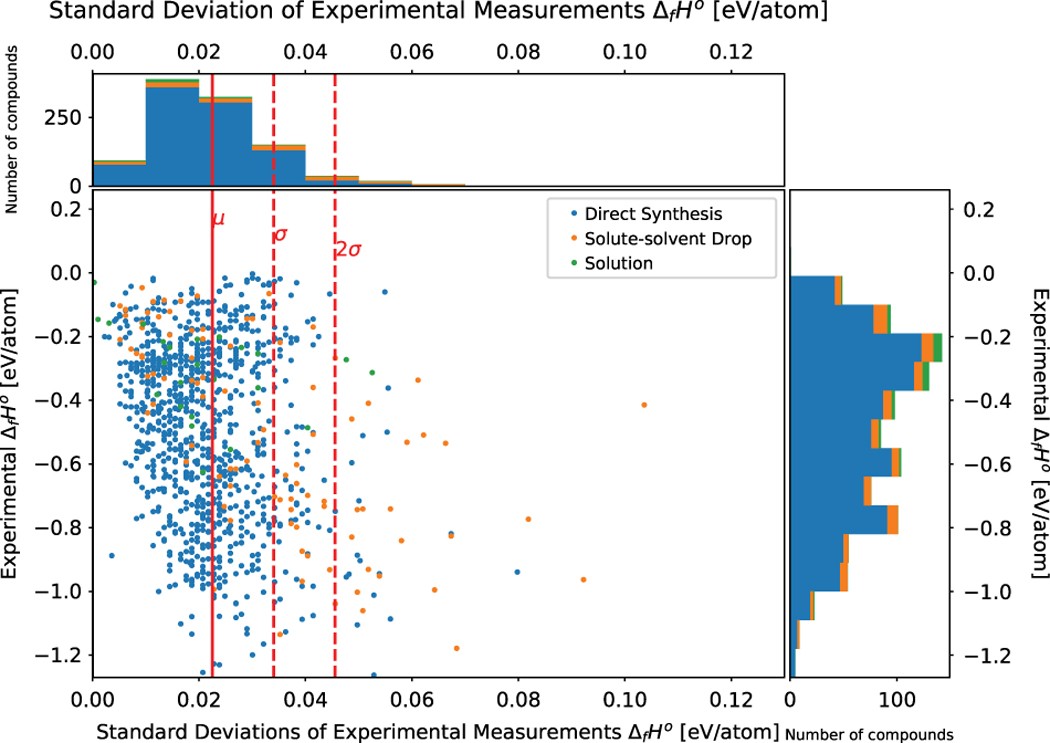
Experimental formation enthalpies for intermetallic phases and other inorganic compounds | Scientific Data

Estimation and uncertainty analysis of standard enthalpy of formation in the liquid state by third-order-group-contribution method - ScienceDirect